Rapid Enrollment
First global site to enroll three patients in a Phase III colorectal cancer trial, accelerating recruitment and study momentum.

Flourish Research delivers high-performance oncology clinical trials across solid tumors and hematologic malignancies, with expertise in complex oncology studies and early-phase trials.
Our wholly-owned oncology sites support sponsors across Phases I–IV, including first-in-human studies, dose escalation trials, and biomarker-driven protocols where patient safety and operational precision are critical.
Flourish teams have experience conducting complex oncology clinical trials across early-phase, Phase III, and basket trial designs. Our investigators routinely manage complex protocols, including trials for novel immuno-oncology agents requiring specialized safety monitoring. We support studies across the treatment continuum, from frontline oncology trials to pretreated patient populations on later-line treatments.

Our oncology sites support research across a broad range of solid tumor and hematologic malignancy studies, including lung, pancreatic, colorectal, breast, prostate, ovarian, melanoma, and multiple myeloma.
Flourish investigators have experience evaluating a wide range of oncology therapeutics, including cytotoxic, targeted, and immune-based therapies. Our teams have supported studies involving advanced immunotherapy approaches such as bispecific T-cell engagers, STING agonists, and adoptive cell therapies, with clinical teams experienced in associated safety monitoring.


Supporting recruitment across both frontline and later-line treatment settings, Flourish Research oncology sites are supported by strong physician relationships and referral networks that enable rapid identification of eligible patients. Our referral ecosystem encompasses more than 200,000 patients and includes 150+ physicians through Central and Southern California.
Typical recruitment snapshot: 24-hour referral response, three days to patient visit, and treatment within two weeks.
First global site to enroll three patients in a Phase III colorectal cancer trial, accelerating recruitment and study momentum.
Recognized by the sponsor as a "Protocol Adherence Champion" for maintaining zero protocol deviations throughout the study.
For an NSCLC program, we were the highest-enrolling site worldwide and supported the study from Phase I through Phase III, enabling a seamless transition and helping bring the therapy to market in under two years.
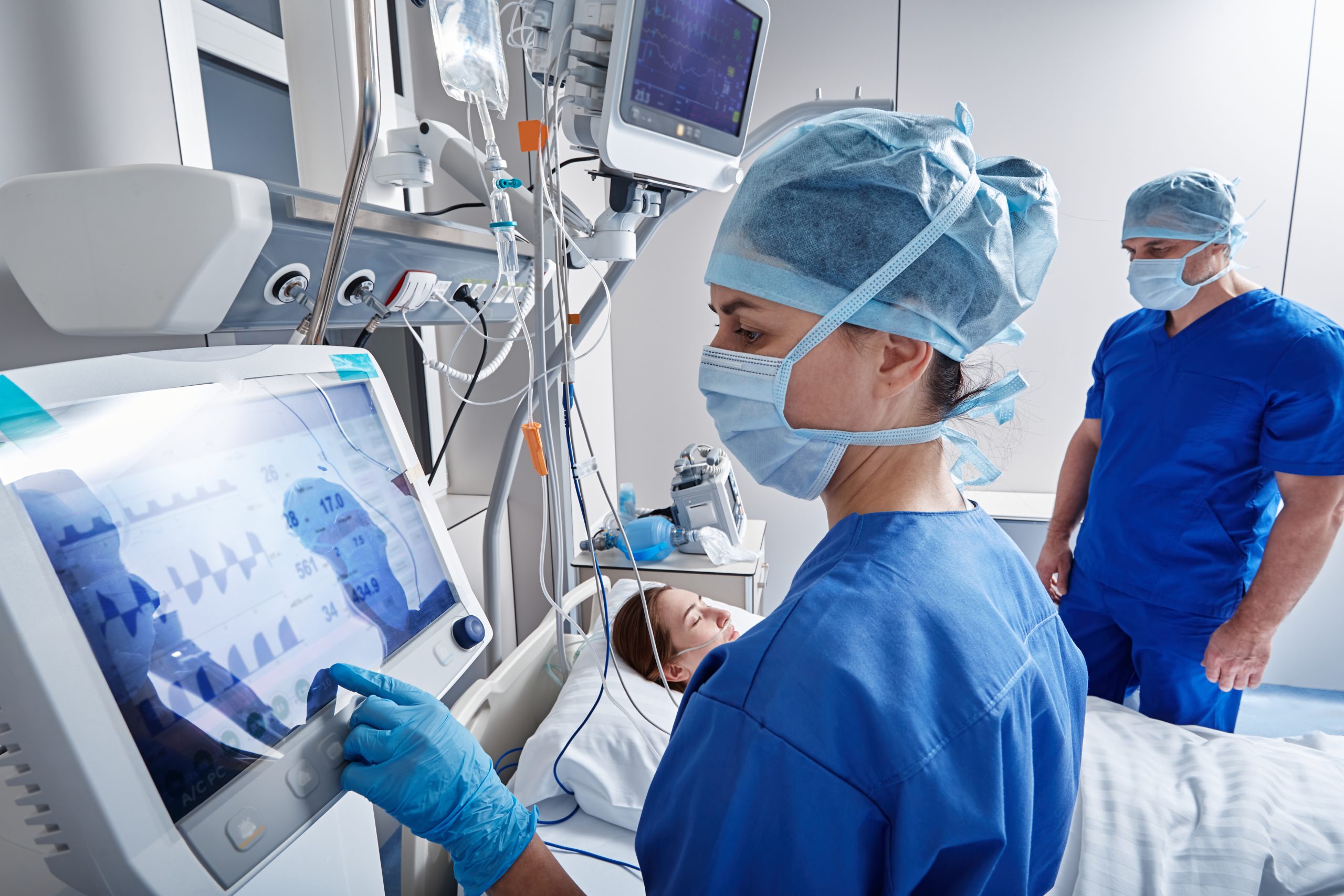
Flourish oncology capabilities are designed to meet the needs of complex trials while safeguarding patient safety.
ICU-Connected Clinical Research Unit
One of our clinical research units is directly connected to a hospital ICU, enabling immediate escalation of care when required to support patient safety during high-acuity oncology trials.
Key features include:
This infrastructure enables safe execution of:

Oncology trials require strict compliance standards. Our sites maintain:
We prioritize data integrity, patient safety, and sponsor transparency throughout the study lifecycle.

Principal Investigator, Founder of Valkyrie Clinical Trials

Study Nurse
Whether you are launching an early-phase oncology study or expanding a late-phase program, Flourish Research provides the operational strength, patient access, and therapeutic expertise to support your clinical development goals. Contact our team today to discuss your upcoming oncology protocol.
Stay current with the latest clinical trial news and learn how Flourish Research is supporting the advancement of life-changing therapeutics.